Emerging technology for verifying engineering controls
The veriDART™ solution leverages patented DNA-tagged tracer particles that safely mimic aerosol mobility and exposure.
Image courtesy of SafeTraces Inc.
How do health facilities professionals verify that indoor air and airflow are safe? Facing a once-in-a-century airborne pathogen like SARS-CoV-2, the answer to this fundamental question has never been more important. They sometimes don’t know the answer because they cannot accurately quantify exposure risk and verify engineering controls in real world spaces. However, emerging technology is rapidly addressing this gap.
Three critical facts
Scientific, medical and public health experts have arrived at consensus on three critical facts over the course of the COVID-19 pandemic:
- SARS-CoV-2 is transmitted via airborne droplets and aerosols — a fact not universally acknowledged in the early stages of the pandemic.
- People in indoor environments are exposed to significantly greater infection risk than in outdoor environments, with enclosed areas, prolonged exposure and poor ventilation representing high-risk factors.
- Engineering controls are critical to mitigating exposure risk, particularly ventilation and filtration.
Health care facility design prioritizes infection control and prevention via enhanced ventilation capabilities, HEPA filtration and negative pressure control. Nevertheless, the pandemic has placed unprecedented stress on these systems due to the high transmissibility of the virus and surges in infections overwhelming designated infection control areas and forcing non-designated areas to handle overflow.
Consequently, mechanical engineers face a complex challenge in verifying the efficacy of engineering controls in designated infection control areas, and responding to the need for rapid repurposing and/or retro-commissioning of areas not originally designed for infection control.
Traditionally, mechanical engineers employ a variety of applied and theoretical methods for verifying efficacy of engineering controls. For verifying ventilation efficacy, tracer-gas based methods using sulfur hexafluoride or carbon dioxide monitors, and direct measurement approaches using instruments, like thermal anemometers and balometers, are common practice.
For verifying filtration efficacy, particle-based methods like dispersed oil particulate for testing HEPA filter integrity and other mostly solid-matter challenge agents for sub-HEPA level filters are common practice. Frequently, non-applied methods augment applied methods like computational fluid dynamic modeling, volumetric airflow calculations and verification of product specifications for HVAC equipment.
In February 2021, the American Industrial Hygiene Association released the “Joint Consensus Statement on Addressing the Aerosol Transmission of SARS-CoV-2 and Recommendations for Preventing Occupational Exposures,” emphasizing the need for “(1) effective, (2) simple-to-use and (3) inexpensive ventilation assessment tools.” Implicit in this recommendation is that existing assessment tools fail to satisfy these three criteria.
From an efficacy standpoint, existing assessment tools have two major drawbacks: (1) They are not liquid aerosol-based and therefore not optimal for verifying ventilation and filtration efficacy for infectious aerosols like SARS-CoV-2, influenza and tuberculosis; and (2) none provide a combined assessment of ventilation and filtration efficacy within one test, requiring triangulation between multiple methods to verify engineering controls holistically.
Beyond efficacy, many practitioners do not consider existing solutions simple-to-use and/or inexpensive, resulting infrequent verification. Consequently, the baseline understanding of airflow, and ventilation and filtration efficacy in real world health care facilities is less robust than one would like. Moreover, the uniqueness of each facility, dynamic nature of airflow and infection risk within facilities, and natural wear and tear of mechanical and HVAC systems, underscores the importance of regular, ongoing verification that exceeds the capabilities of existing assessment tools.
Technical advances
However, the pandemic has spurred groundbreaking technological advances that have pushed us closer to effective, simple to use and inexpensive solutions that can be used routinely in real world spaces.
As one example, SafeTraces Inc., Pleasanton, Calif., has developed the first commercially available aerosol-based diagnostic solution for verifying ventilation and filtration efficacy for airborne pathogens. Developed with support from the National Institutes of Health and technical experts at leading research universities like Stanford and MIT, the veriDARTTM solution by SafeTraces leverages patented DNA-tagged tracer particles that safely mimic aerosol mobility and exposure to identify high-risk infection hotspots and transmission routes, regularly assesses HVAC system performance at an affordable price point and ease of use, and informs remediations with a rigorous science-based, data-driven methodology.
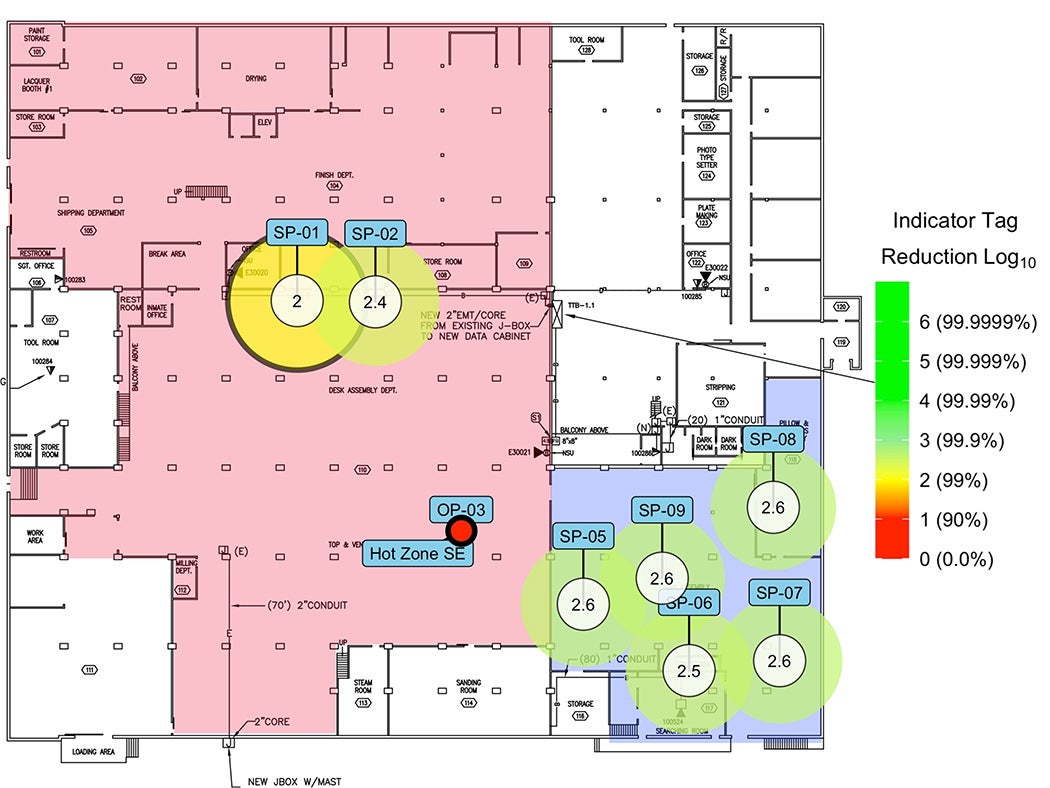
The veriDART™ by SafeTraces was used to iteratively verify negative pressure controls and airflow isolation between hot zone and safe area at a major superspreader site.
Images courtesy of SafeTraces Inc
SafeTraces’ technology consists of two main components.
First, the particle technology enables the SafeTraces team to safely simulate the chemical composition, particle size distribution and detection method of virus-laden respiratory droplets and aerosols to verify ventilation and filtration efficacy in one test. DNA provides three significant advantages relative to existing methodologies: (1) DNA is highly sensitive to detection, meaning relatively small amounts of DNA can be used to test large areas at very precise levels; (2) unique DNA identifiers can be tagged to different tracer solutions, meaning multiple tracers can be released simultaneously simulating viral emissions at different points in a facility and each sample point can be tested against each unique tracer to develop a sophisticated assessment of aerosol mobility and exposure patterns; and (3) the DNA-tagged tracer solutions are completely safe, using food-grade, water-soluble materials that are FDA-Generally Recognized as Safe (FDA-GRAS), that are OSHA- and NIOSH-compliant, and that allow for occupants to be in the facility during testing.
Second, the data technology enables the SafeTraces team to precisely measure DNA signal strength, correlate test results to a relative exposure risk scale informed by infectious disease science and generate heatmaps and other visualizations to clearly communicate key findings and implications.
In recent months, SafeTraces has supported a diverse set of corporate, commercial real estate and government clients, that have integrated the veriDART solution into critical health, safety and financial decision-making processes and ongoing environmental health and safety programs.
Notably, SafeTraces has supported health care facilities in three main areas: (1) It verified the efficacy of engineering controls in designated infection control areas, verifying ventilation, filtration and negative pressure control performance against reference standards like American National Standards Institute/ASHRAE/American Society for Health Care Engineering's Standard 170, Ventilation of Health Care Facilities; (2) it verified the efficacy of engineering controls in repurposed and/or retro-commissioned areas to improve building operation and maintenance procedures, often iteratively testing ventilation, filtration and negative pressure controls until a level of performance was achieved to ensure adequate infection control; and (3) it verified the efficacy of engineering controls in non-infection control areas, including high-risk areas like lobbies, waiting rooms and breakrooms.
Real-world examples illustrate the practical value of SafeTraces’ verification solution for infection control.
In late 2020, veriDART supported the retrofit of an existing long-term care facility resident wing into a temporary isolation unit, with modifications including increased ventilation rates, negative pressure controls, HEPA filtration and physical airflow barriers. The test results quantified the relative impact of varying ventilation rates, negative versus neutral airflow modes and other critical priorities, such as the “blast radius” of particle dispersion and particle detection levels in the HVAC system and filters to inform critical design decision-making.
In a separate engagement, SafeTraces supported a large prison during a major coronavirus outbreak that infected hundreds of inmates and staff. The crisis forced the prison’s leadership to evacuate the main prison complex and establish a large makeshift infection ward in a vacant warehouse on its premises. Prior to populating the ward, the prison’s leadership sought to verify airflow isolation and efficacy of negative pressure controls between the “hot zone” designated to house hundreds of infected inmates and the “safe area” designated for medical and administrative staff. The SafeTraces team worked with the prison’s facilities team to iteratively assess performance of negative pressure controls, with dramatic improvement between initial and final results and ultimately mitigating further viral spread (see graphic).
Quantifying risk
Emerging technologies are transforming the ability of health care facilities to quantify exposure risk and verify engineering controls for airborne pathogens with far-reaching consequences beyond the current pandemic. The benefits of these technologies-enabled solutions are enormous: enhancing patient and provider safety; regularly auditing HVAC system performance in ways never before possible; targeting remediation spending to its highest value opportunities; supporting compliance for rapidly evolving regulation; and eventually factoring into liability insurance and other mechanisms for evaluating and underwriting risk. It will be important for facilities engineers to be adaptive and consider how and where emerging technologies can best be integrated into their respective facilities for improved infection control and safety.
Erik Malmstrom is CEO of SafeTraces Inc., Pleasanton, Calif. For more information, readers should contact info@safetraces.com or visit www.safetraces.com.




