Alignment of standards for facility ventilation
The operating room is designed as one of the most restrictive clinical environments where invasive procedures are performed.
Image ©Jeff Goldberg/Esto and courtesy of NBBJ
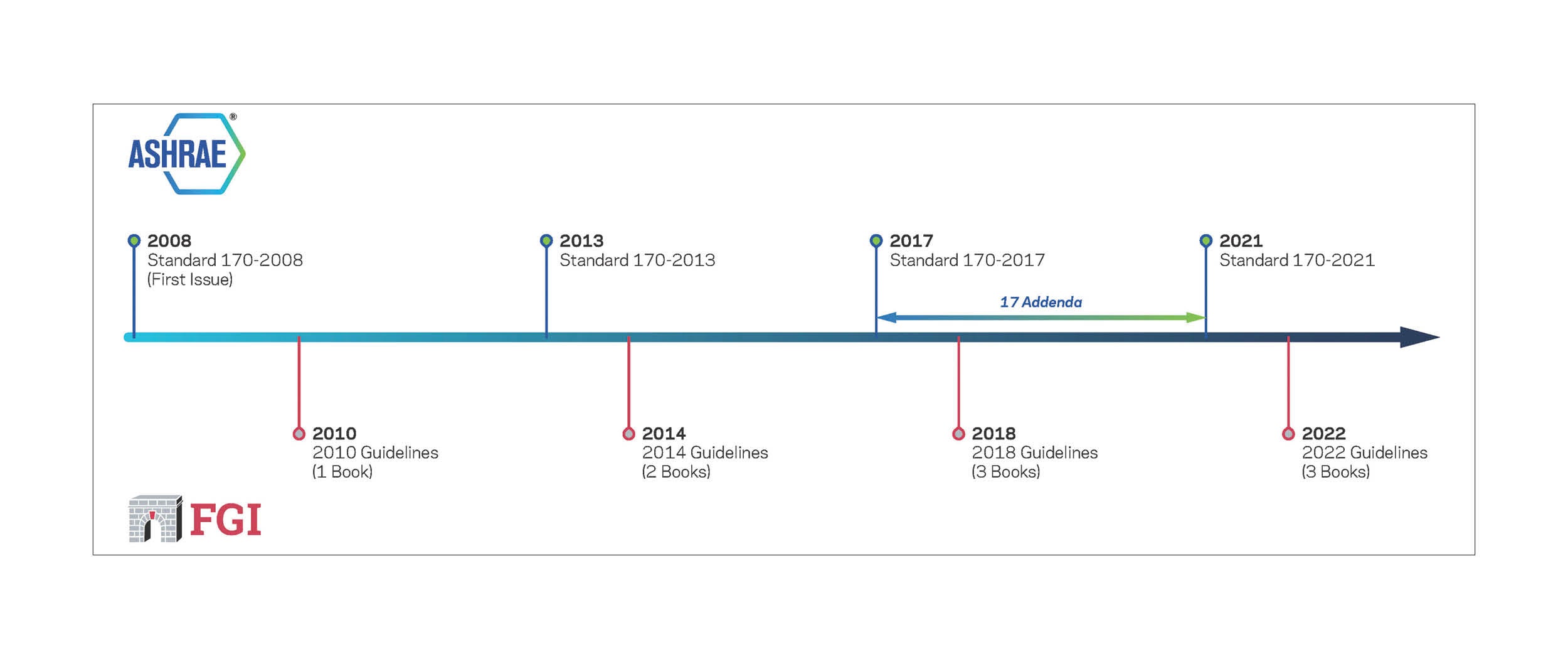
ASHRAE and the Facility Guidelines Institute (FGI) have collaborated since 2008 on coordinating standards for ventilation of health care facilities, in conjunction with the American Society for Health Care Engineering (ASHE).
Beginning with the 2010 FGI Guidelines for Design and Construction of Health Care Facilities, ANSI/ASHRAE/ASHE Standard 170, Ventilation of Health Care Facilities, has been included in the Guidelines documents. The recently released 2022 edition of the FGI Guidelines references and includes the 2021 edition of Standard 170.
FGI, ASHRAE and ASHE have worked together to better align their documents with a goal of making it easier for health care designers and facilities managers to use them in planning, designing and constructing health and residential care facilities.
Content development
Since 2008, members of FGI’s Health Guidelines Revision Committee (HGRC), the interdisciplinary body responsible for development of Guidelines content, have brought their expertise to ASHRAE’s Standing Standard Project Committee (SSPC) 170, a body that has consistent ASHE representation and is responsible for development of Standard 170, which ASHE co-publishes.
The resulting collaboration enabled the transition of all ventilation requirements into Standard 170 for the 2022 FGI Guidelines and Standard 170-2021.
The three FGI Guidelines documents (Hospital, Outpatient and Residential) provide minimum functional program, risk assessment, space, architectural detail, surface, built-in furnishing and building system requirements for clinical and support areas of hospitals, ambulatory care facilities, rehabilitation facilities, and nursing and other residential care facilities. The Guidelines is updated every four years to stay current with changes in the field and to provide clarifications that can ease adoption and compliance.
Although the documents are called “guidelines,” they are written as fundamental standards and become enforceable when states or federal agencies adopt or reference them in state laws, codes, rules or regulations. The Joint Commission, Healthcare Facilities Accreditation Program, DNV, many federal agencies and authorities in 42 states use the Guidelines as a code or a reference standard when reviewing, approving, surveying, licensing, certifying or accrediting newly constructed or renovated facilities. Other states reference or extract language from the Guidelines for information and when developing their own codes.
Standard 170 provides ventilation system design requirements that support environmental control for health care facilities. These address multiple aspects of ventilation, including control of odor, temperature and humidity. Standard 170 establishes minimum engineering design criteria for heating, ventilation and air-conditioning systems for patient care areas and related areas in health care facilities. The document was developed in 2008 to provide a consistent source of guidance on ventilation for the health care industry, with the idea that an effective, sensible ASHRAE standard could be readily adopted into other standards.
Resources
Standard 170 is under the category ASHRAE terms “continuous maintenance” project status. The maintenance committee — SSPC 170 — comprises a mixture of members of FGI’s HGRC, ASHE, the Association of Professionals in Infection Control and Epidemiology, and ASHRAE who are experienced in health care facility ventilation. The committee meets periodically to apply its broad expertise in health care environments to the task of keeping Standard 170 current with practices in the field, developing changes that — after a public review process — are published as official changes (addenda) to the current document. These official addenda are available for download from the ASHRAE, FGI and ASHE websites. It is the hope of SSPC 170 that state and federal agencies will adopt these addenda as they are issued because they represent state-of-the-art thinking in the industry.
Major changes
Significant improvements were made to Standard 170 for the 2021 edition, most notably the conclusion of efforts to distill the standard into three distinct sections: hospital, outpatient and residential. As a result of this change, the updated standard follows the same framework as the 2018 Guidelines, which was published as three books — one each for hospitals; outpatient facilities; and residential health, care, and support facilities.
This discussion of the revisions and updates that appear in the 2022 FGI Guidelines and Standard 170-2021 is organized by the major components of Standard 170. Although an initial version of Standard 170-2021 was published midyear 2021, the version published in the 2022 Guidelines is Standard 170-2021 (with Standard 170-2021 addenda c and d and errata).
- Glossary and definitions. In the front matter of both the Guidelines and Standard 170 is a section where commonly used words and terms are defined. FGI refers to this as the “glossary,” and ASHRAE as “definitions.” For the 2022 Guidelines and Standard 170-2021, the respective sections have been revised and correlated. Words that appear in both documents — including procedure, operating, and Class 1, 2 and 3 imaging rooms — are now defined similarly.
A significant change in language is the replacement of “invasive fluoroscopy” with “procedural fluoroscopy” to avoid confusion with the term “invasive procedure.” FGI and ASHRAE agreed the term “invasive fluoroscopy” led to confusion, as a room where this procedure is performed can be designed mechanically similarly to a procedure or Class 2 imaging room, while a room where an invasive procedure will take place must be designed as an operating or Class 3 imaging room. As well, the term “invasive imaging procedure room” was removed from the Standard 170-2021 definitions.
- General requirements. Paragraph (a)(2) in the Standard 170 “General Requirements” text for hospital, outpatient and residential facilities (sections 7.1, 8.1 and 9.1, respectively) provides language to address ventilation for those spaces located in a health care facility that are not explicitly specified in the standard’s design parameter tables. The new phrasing acknowledges that space ventilation requirements may exist in other codes and standards for these spaces:
7.1 General Requirements. (a)(2)
The ventilation requirements in this table are intended to provide for comfort as well as for asepsis and odor control in spaces of a health care facility that directly affect patient care. For spaces not specifically listed here, ventilation requirements shall be that of functionally equivalent spaces in the table. If no functionally equivalent spaces exist in the table, ventilation requirements shall be obtained from ASHRAE Standard 62.1 in the absence of other codes or standards that govern those space ventilation rate requirements. Where spaces with prescribed rates in both Standard 62.1 and Table 7-1 of this standard exist, the higher of the two air change rates shall be used. [Bold added for emphasis]
- Use of inhalation anesthesia. In the general requirements sections for hospital and outpatient facility ventilation, it has been clarified that the type of anesthesia employed for a procedure does not automatically demand a specific type of room or space. Or, put more bluntly, the use of inhalation anesthetic gas can occur in any type of room and does not need to occur in an operating room setting.
Although the HGRC worked extensively to clarify the definition of “invasive procedure” in the 2018 Guidelines and to link the level of invasiveness of a procedure with the determination of room classification, some contradictory language remained in Standard 170-2017.
The new, clarifying language in Standard 170-2021, which appears in Section 7.1(a) and Section 8.1(a) of the document, states the following:
7.1 General Requirements. (a)(7)
Unless a higher ventilation rate is stipulated in Table [7-1 or 8-1] or elsewhere in this standard, wherever anesthetic gases [e.g., nitrous oxide, xenon, halogenated volatile anesthetic agents] are administered outside of an operating room, procedure room, or Class 2 and Class 3 imaging rooms, ventilation shall be provided at a minimum rate of 2 outdoor air changes per hour and 6 total air changes per hour. (Informative Notes: [1] Refer to NFPA 99 [2021] in Informative Appendix E for WAGD piping and gas scavenging requirements. [2] “Anesthetic gases” commonly refers to nitrous oxide and xenon but may also include halogenated volatile anesthetic agents such as desflurane, sevoflurane, and isoflurane.) [Bold added for emphasis]
- Airborne infection isolation (AII) rooms. In response to the many challenges organizations have faced during the pandemic, Standard 170 more readily accommodates, via new Paragraph (b)(2) in Section 7.2.1, the varied conditions that arise in planning, designing and implementing AII rooms, especially in retrofit situations, by permitting flexibility in exhaust discharge arrangements from these spaces in alignment with the Centers for Disease Control and Prevention guidelines. The requirement for all air from AII rooms to be exhausted directly outdoors has been replaced with the following language:
7.2.1 Airborne Infection Isolation (AII) Rooms. (b) ...
All exhaust air from the AII rooms, associated anterooms, and associated toilet rooms shall be discharged by one of the following methods:
1. Discharged directly to the outdoors without mixing with exhaust air from any other non-AII room or general exhaust system.
2. Discharged into the general exhaust stream, provided the AII exhaust air first passes through a HEPA filter. The HEPA filter, including ductwork and fans, shall be under negative pressure (suction side) for any supplemental fan used to account for filter pressure drop, and all exhaust ductwork shall be kept under negative pressure in accordance with Section 6.3.2.1. [Bold added for emphasis]
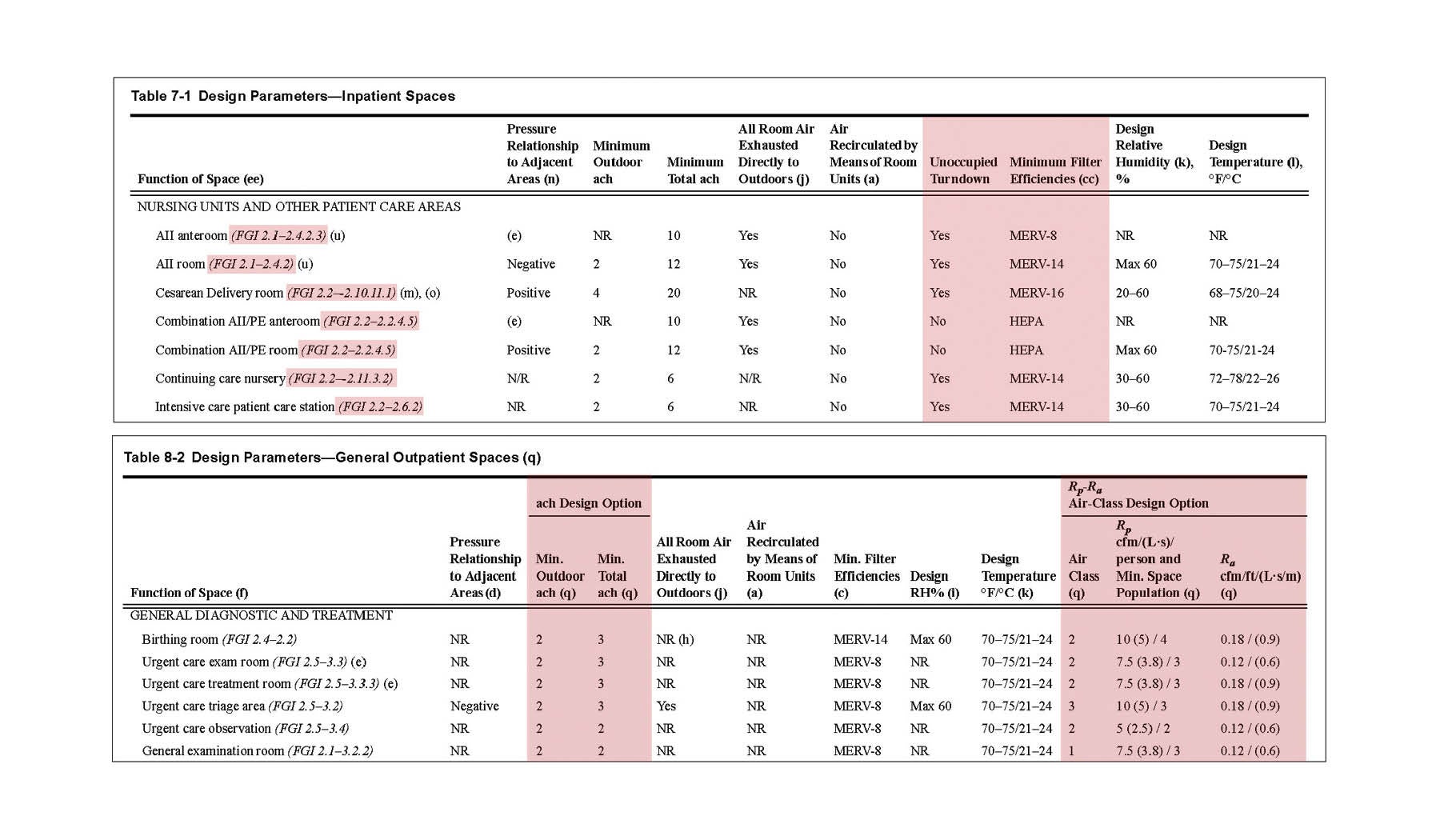
- Design parameter tables. Several updates and improvements have been made to the ventilation tables in Standard 170-2021 to make the information easier to access, providing more information in one location and cross-referencing space requirements in the Guidelines. In the “Function of Space” column in each table, space names are aligned with the 2022 Guidelines and include the appropriate Guidelines section number. The tables have been reorganized with more subheads for ease of use, and spaces that do not apply to the specific facility type (hospital, outpatient or residential) have been removed from those tables.
In addition to these refinements, two new columns have been added to the design parameter tables in the document. A column titled “Unoccupied Turndown” has been created to indicate spaces where this practice is acceptable. Sections 7.1(a)(3) for hospitals and 8.1(a)(3) for outpatient facilities explain what is allowed when implementing unoccupied turndown:
7.1 General Requirements. (a)(3) ...
Except where indicated by a “No” in the “Unoccupied Turndown” column, the number of air changes shall be permitted to be reduced, and temperature and design relative humidity altered, when the space is unoccupied, provided that the required pressure relationship to adjoining spaces is maintained while the space is unoccupied and that the minimum number of air changes, temperature, and design relative humidity indicated are reestablished anytime the space becomes occupied. [Bold added for emphasis]
The other new column, titled “Minimum Filter Efficiencies,” moves the filter requirements from Table 6.4 into Tables 7-1, 8-1, 8-2 and 9-1 in Standard 170-2021. This change clarifies filtration requirements on a space-by-space basis, offering flexibility so users can apply filtration selections that most rationally meet their goals for first, operational and energy costs. Filtration requirements were increased in some spaces and reduced in others, with the net effect of maintaining safe environments for occupants (see the table excerpt on this page).
For outpatient facilities only, a second design parameter table has been added to support an alternative design option for calculating ventilation. Table 8-1 is for “specialized outpatient spaces,” aligning with FGI content addressing the acute care outpatient spaces commonly found in free-standing emergency facilities, outpatient surgery centers, imaging centers, endoscopy facilities and infusion centers.
A new Table 8-2 is for “general outpatient spaces,” addressing non-acute care environments such as general outpatient medical facilities, urgent care, physical therapy, behavioral and mental health care, and birth centers. In lieu of calculating ventilation via air change rate (ACH), the optional path allows calculation of the ventilation rate using provided values for cubic feet per minute (cfm) per person and cfm per square foot, and air class (see the table excerpt on this page).
Table 9-1, in the updated “Residential Health, Care, and Support Spaces” section of Standard 170, now clearly and more comprehensively details the design parameters for ventilation of nursing home and hospice facility spaces and for assisted living facilities. Also included in the table are service areas and support areas in these facility types.
Looking ahead
Standard 170 is already on the move, with SSPC 170 having issued an addendum draft for public review and seeking comments on whether to allow natural ventilation for a variety of spaces.
Similarly, an addendum draft has been issued to consider adjustments to how the outdoor air component of the air change rate is calculated to improve clarity of methodology and better coordinate with ASHRAE 62.1, Ventilation for Acceptable Indoor Air Quality, the standard that applies to spaces in health and residential care facilities other than patient care areas, resident care areas and related support areas.
Beyond the day-in and day-out efforts of the SSPC in refining existing content, coordinating content with FGI and identifying improvements arising from changing technologies, the committee has taken an active role in prioritizing and encouraging the development of ASHRAE research and further research partnerships with others in the industry.
Key research that melds environmental and clinical space factors can inform committee decision-making that supports safe adjustments to the standard, but this work doesn’t happen without planning to gather the right information or funding to realize the results.
As organizations face pressures to implement energy efficiency and decarbonization, the role of air quality and quantity will be looked at with increased scrutiny. SSPC 170 is working hard to be a supportive player in this quest.
Key groups behind Standard 170
A collaborative effort of ASHRAE and the American Society for Health Care Engineering (ASHE) that is accredited by the American National Standards Institute (ANSI), ANSI/ASHRAE/ASHE Standard 170, Ventilation of Health Care Facilities, delivers critical guidance for designers and operators of these front-line facilities. Here’s a closer look at each of the organizations’ roles:
ANSI. This is the entity that confirms that the standard has met specific criteria to be nationally accredited and ready for adoption by regulators. While ANSI itself is a 501(3)(c) private nonprofit organization, its role is integral in serving as the administrator for the voluntary standardization system in the U.S., guiding many standards-developing organizations as they develop and improve their standards.
ASHRAE. This is a 51,000-member, volunteer-led organization focused on education, research and service to the HVAC industry, including development of technical standards and guidelines.
ASHE. This organization has been a co-sponsor of ASHRAE Standard 170 since its inception. ASHE is a professional membership group of the American Hospital Association (AHA) with more than 12,500 members. Core to the role that ASHE provides in the partnership with ASHRAE are:
- Insights from ASHE members who experience Standard 170 through operational practices and regulatory reviews. ASHE has had consistent representation on the Standard 170 committee, ensuring open communications between its members and ASHRAE’s Standing Standard Project Committee (SSPC) 170. As well, ASHE organizationally approves all SSPC 170 publications, including addenda, prior to release.
- The advocacy that ASHE and AHA provide for adoption of a coordinated regulatory framework includes ASHE’s participation on SSPC 170; its work in coordinating the health care-related standards of various entities, including ASHRAE, the National Fire Protection Association, International Code Council and the Facility Guidelines Institute; and its encouragement of the Centers for Medicare & Medicaid Services to adopt updated rules.
About this article
This feature is one of a series of articles published by Health Facilities Management in partnership with the American College of Healthcare Architects.
Bryan Langlands, FAIA, FACHA, is a principal with NBBJ, and Michael Sheerin, PE, LEED AP, is chief executive officer at TLC Engineering Solutions. They can be reached at blanglands@nbbj.com and michael.sheerin@tlc-eng.com.