What’s new for hospital airflow?

Addendum e of Standard 170-2017 indicates that an owner or facility manager of a health care facility shall provide an operational plan in the event of an extended power or fuel outage.
Image by Kittner Photography and courtesy of TLC Engineering Solutions
Under normal circumstances, the devoted efforts of state health authorities, health facilities professionals, and affiliated industry partners involved in health care codes and standards consists of mad dashes to airports, late nights poring over thick books and the occasional social gathering with peers from around the country who share the desire to contribute insights and advance these documents to the next level.
But this past year has brought many changes. On one hand, traveling was set aside, ushering in a new era that meant joining in was as easy as plugging in from a desk or home office. On the other hand, every day seemed to present an emergency condition or new calamity — whether fires, floods or hurricanes — and even the most dutiful volunteer code writer understandably begged for a break at one point or another during the year.
Amid this backdrop, two extraordinary truths arose for the committee charged with the 2021 edition of the American National Standards Institute/ASHRAE/American Society for Health Care Engineering (ASHE) Standard 170, Ventilation of Health Care Facilities: The standard survived a pandemic, and the standard was prepared for its next edition. Vigorous discussion and countless pre-publication research reports were studied to assess if key components of Standard 170 would need immediate change to ensure safety, and they never arose.
There are a variety of better and best practice measures that the Standard 170 committee is assembling into guidance that may constitute a future white paper, but the core elements and strictures of ventilation for infection control have weathered the year as well as any other aspect of the environment of care.
Perhaps because the year presented fewer social distractions, the standard was prepared for republication in time for incorporation into the Facility Guidelines Institute (FGI) Guidelines. The committee worked through lingering addenda and completed a few new ones to firm up content. This summary is meant to inform health facilities professionals regarding the latest changes that will be published in the 2021 edition of Standard 170, and the topics of future changes being studied.
Recent addenda
In Addendum m, the designation “psychiatric” was updated to “behavioral and mental health” throughout the standard in coordination with industry trends and FGI updates, and the placement of HVAC systems and their access was made subject to consideration in the patient safety risk assessment. While it is common practice based on the seminal design resource Behavioral Health Design Guide (bhfcllc.com/design-guide), the addendum ensures that the standard is now explicit that seclusion, restraint and observation rooms are required to be equipped with air grilles that are secure and ligature-resistant for the safety of patients and residents in these spaces, and this guidance is applied consistently across the chapters for hospital, outpatient and residential facilities.
Addendum j was a significant change, as it made considerable updates to Chapter 8 on outpatient facilities. While it may strike outside observers that the work was changing recent modifications created by Addendum n, they must appreciate that Addendum n was started before the publication of the 2017 edition and that the content of Addendum n simply started the edit process to move chapters 7, 8 and 9 into the realm of their distinct occupancy use types (i.e., hospital, outpatient or residential health). Specifically, Addendum j reflects the committee’s more advanced deliberations and conclusion that the intent of Standard 170 is to be the cognizant standard to address health care spaces in any type of facility that they may be housed.
To preserve the appropriate acute care requirements with the rigor that is expected of those spaces, these spaces were aggregated into Table 8.1, the ventilation requirements for spaces in the following specialized outpatient facility types: outpatient surgical, endoscopy, infusion, renal dialysis, freestanding emergency departments, and imaging facilities with Class 2 and 3 imaging rooms, which is consistent with the guidance in the FGI outpatient volume.
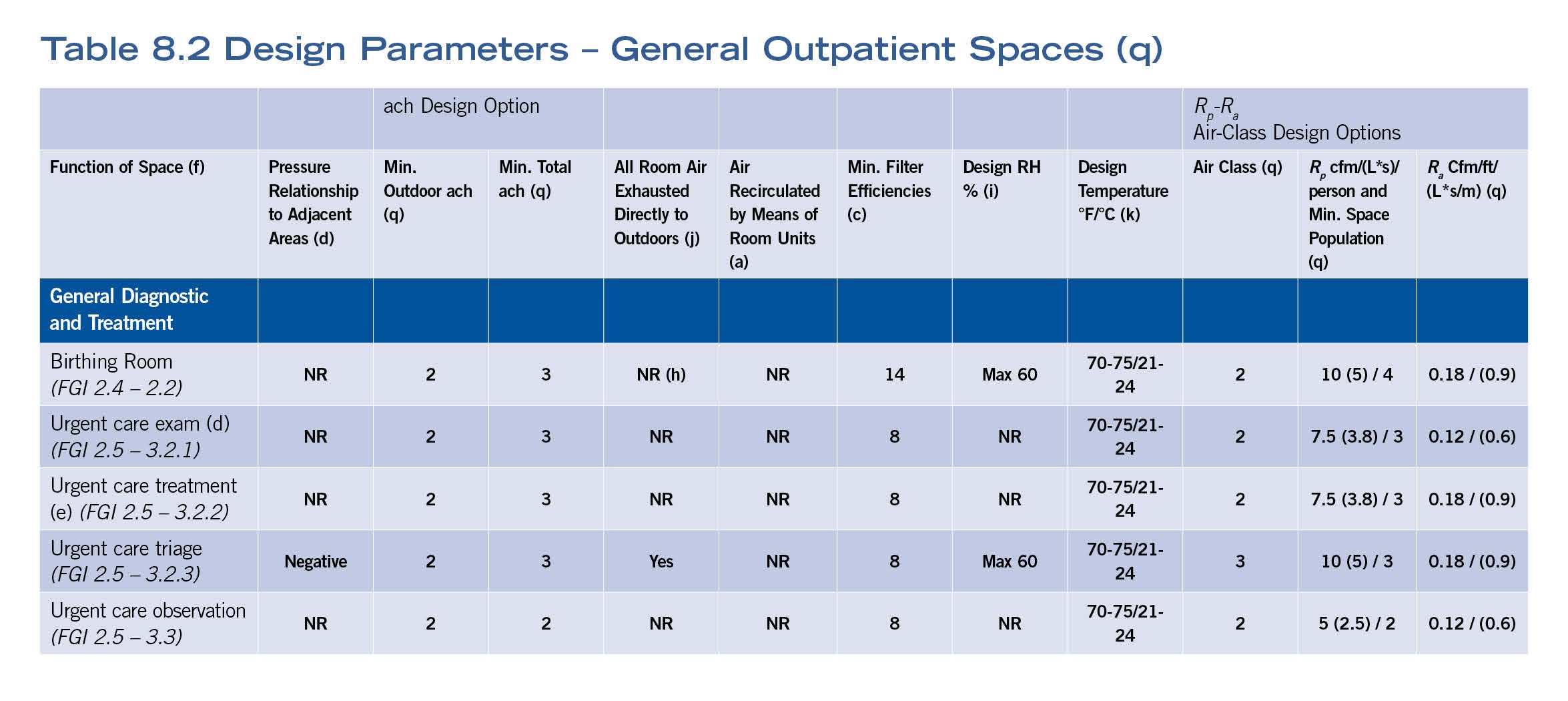
More inventiveness was necessary to accommodate the varied occupancy conditions that exist outside of a pure health care environment, so the committee created a separate Table 8.2 that listed “General Outpatient Spaces” for this range of spaces (see table above). Examples include urgent care exam rooms, Class 1 imaging rooms and occupational therapy. These are spaces that obviously can occur in a wide swath of health care and health care-affiliated and -adjacent settings.
This work coincided with and coordinated with Addendum af of ASHRAE Standard 62.1, Ventilation for Acceptable Indoor Air Quality. In a first for this standard, compliance can be accomplished via the air change method or an optional path that utilizes the “Rp” and “Ra” method. This allows designers the option of choosing the path that best suits their needs, given the different calculation methods that are used and the different users that may engage with the standard for these types of spaces.
Addendum l (lower case “L”) arises out of the evolving surgical environment and its increasing integration with technology. Working with members of the FGI Health Guidelines Revision Committee’s (HGRC’s) surgical/imaging expert team, the committee revised and added key definitions regarding invasive procedure and hybrid operating rooms (ORs), and aligned with FGI in reference to the new Class 1, 2 and 3 imaging room categories.
Addendum l also incorporates an important change related to the use of anesthetic gases that was recognized by this group and a frequent question from those seeking interpretation requests of the committee. Previously, the use of anesthetic gases necessitated that the ventilation of the space complies with OR parameters. There are notable instances where anesthetic gases are used as an analgesic or anxiolytic to reduce the stress a patient may experience during a diagnostic imaging procedure, in lieu of full sedation as might occur for an invasive procedure. Thus, a direct correlation cannot be made based on the usage of anesthetic gases alone and the level of ventilation requirements within a room. In working with clinical experts from FGI, the use of anesthetic gases was de-linked from OR ventilation parameters and allowed to occur where needed with the caveat that the space must meet at least six total air changes and two outdoor air changes.
Finally, while Addendum a widely updated the type and placement of filtration, and notably brought the shift to having columns in the ventilation tables to define filtration requirements by space (along with a new turndown column), importantly, Addendum l revised to HEPA filtration levels and the placement of the filtration in the air terminal device for specific operating and for Class 3 imaging rooms, as noted in item c:
7.4.1 Operating Rooms, Operating/Surgical Cystoscopic Rooms, and Caesarean Delivery Rooms, and Class 3 Imaging Rooms. These rooms shall be maintained at a positive pressure with respect to all adjoining spaces at all times. A pressure differential shall be maintained at a value of at least +0.01 in. wc of water (2.5 Pa). Each room shall have individual temperature control. These rooms shall be provided with a primary supply diffuser array that is designed as follows:
[…]
c. In operating rooms or Class 3 imaging rooms designated for orthopedic procedures, transplants, neurosurgery, or dedicated burn unit procedures, HEPA filters shall be provided and located in the air terminal device.
The committee continues to evaluate the impact of the pandemic on ventilation system arrangements and how the standard supports the varied approaches that have been needed. Notwithstanding the emergency condition nature of some arrangements, the standard fared well under that scrutiny.
One issue that was highlighted by feedback and questions related to airborne infectious isolation rooms and general exhaust systems. This is especially helpful in expedited situations and retrofit conditions as were experienced under the pressures of the pandemic and also frequently faced in older buildings with fixed glazing.
The committee promptly reviewed and agreed that the HEPA filtered discharge of the exhaust from these spaces could be routed to the general exhaust, in coordination with Centers for Disease Control and Prevention guidance that has allowed such arrangement. The update is reflected in addendum s.
Previous updates discussed in the January/February 2020 edition of Health Facilities Management provided insights on other addenda, and facilities professionals are encouraged to circle back for information on those changes as well.
Work on the horizon
The committee often shepherds new work forward by assigning committee members and sometimes outside experts into a work group that can study the topic and report.
The committee has formed a work group to study the temperature and humidity ranges that are listed in the tables. While space temperature and humidity ranges are a de facto reference item in the standard, they are fraught with the weighty concerns of another sort: that perhaps accreditation inspectors might make citation of some divergence from the standard.
The standard is clear that — in addition to not being an operational standard — these temperature and humidity ranges are design parameters as opposed to points on a compass. Further, certain spaces are allowed to diverge from the stated range based on surgeon or surgical parameters.
A more comprehensive overview of recent research, along with a contemplative approach to the role that the standard still plays in occupied spaces, will have this work group wrestling to strike the right balance in any changes offered to the full committee. Frequented by engineers and those seeking orderliness, the committee can, on occasion, create and offer some complex equations in defining the paths of compliance. While this can be vexing, it can become confounding when requirements span more than one ASHRAE standard. Then, the designer is challenged with the task of coalescing the layered elements of multiple standards into a cohesive answer.
Such is the case at present as facilities professionals calculate the outdoor ventilation air requirements for health care. While Standard 170 addresses the outdoor ventilation amounts for listed health care spaces; non-health care spaces, such as offices, conference and breakrooms, defer to ASHRAE 62.1, Ventilation for Acceptable Indoor Air Quality.
Recognizing this, the Standard 170 committee has initiated an outside air calculations work group that will work with ASHRAE 62.1 to better inform users of the standard on how to apply the mix of requirements that a typical health care setting may pose. While new equations may be created, the possibility of a more efficient and understandable process is a laudable goal for any work group and well worth the efforts.
The Standard 170 committee also is evaluating a course change. Presently, the Standard 170 document refers to the documents USP General Chapters <795> Pharmaceutical Compounding — Nonsterile Preparations; <797> Pharmaceutical Compounding — Sterile Preparations; and <800> Hazardous Drugs — Handling in Healthcare Settings and sets basic minimum ventilation marks that obviously need to be exceeded to comply with the applicable USP requirements, as all facilities professionals have experienced.
With halting and sometimes delayed changes related to the pharmacy spaces, the Standard 170 committee is reaching out to USP and proposing a more proactive approach by consolidating the pharmacy HVAC requirements within Standard 170. This would offer USP the ability to reference the Standard 170 document and maintain their concentration on the requirements for the safe handling and manufacturing of pharmaceuticals, and less on the physical space requirements within a health care setting.
Additionally, the natural ventilation subcommittee has established a work group to take the considerable knowledge that was applied in the recent update of the related section in ASHRAE Standard 62.1, and some of their participants, to develop draft content that would form a future addendum to address natural ventilation in health care settings. This is a complex matter that will take a while, but starting off on the right foot is certainly helped along by the involvement of the right expertise.
Further research
Great strides have occurred in ASHRAE content when it stemmed from research studies. The same actions have informed and propelled Standard 170 forward quite often, whether it was the OR air diffuser array or the safe applications of chilled beam-type systems in a patient room.
Rick Hermans, the founding chair of Standard 170 and unofficial research historian of the committee, would caution that one research study cannot address every element or circumstance that the standard may have to address on a topic. Therefore, patience is often a remedy to overcoming those gaps, finding and encouraging additional research so the committee may ultimately triangulate the interrelated factors that best address and either affirm requirements in the standard or identify appropriate adjustments.
Working with ASHRAE’s Technical Committee 9.6 for health care facilities, the Standard 170 committee is establishing a spate of research initiatives. These areas of research range from the fundamental matters of determining what contaminants of concern are in the air in a health care setting and how they affect people to the more practical matters of the effective placement of air diffusers in a health care space to create safe areas.
A frequent challenge in research considerations for health care environments is the lack of access often afforded researchers to active clinical settings due to the caution and privacy issues. Thus, we are encouraged by private initiatives such as an OR study that compared ventilation rate and sterile field contaminant rates, as presented to the committee at the ASHRAE Winter Conference last February in Orlando, Fla. They had even collected real-time data during actual procedures. Well-designed studies that meet an academic rigor are a tremendous asset to the public and to the committee as well.
While these requests for research must be approved for funding through ASHRAE or funded through partnerships with Standard 170 partner ASHE or other industry partners such as FGI, they are important topics, and the results could inform and enhance the standard for many years.
Many challenges
Health facilities teams and their partners have overcome many challenges to create safe places for patients and staff during the pandemic. Likewise, this work was often aided by the meaningful collaborations and emergency condition reviews by authorities having jurisdiction.
The Standard 170 committee appreciates that dedicated individuals and organizations continue to offer their expertise toward health care codes and standards in good and difficult times. The wealth of knowledge they share, and the practical experiences they relate, end up guiding the development of this work and enliven the work of committees to do the utmost for them and those they represent.
Finally, the committee offers encouragement to the FGI team as the HGRC continues its efforts to review proposed changes, vote and complete its efforts in preparation for the 2022 editions of the Guidelines. The Standard 170 committee is strengthened by the clinical insights the FGI team provides to the development process.
Michael P. Sheerin, PE, LEED AP, is CEO and chair of TLC Engineering Solutions, Orlando, Fla., as well as a founding member and current chair of the ASHRAE/ASHE Standard 170 committee; and Aaron L. Johnson, PE, LEED AP BD+C, is a managing principal at TLC Engineering Solutions, Orlando, Fla., and a nonvoting member of the ASHRAE/ASHE Standard 170 committee. They can be reached at michael.sheerin@tlc-eng.com and aaron.johnson@tlc-eng.com.